- +61 418 122 641
- Info@medicaldevicesponsors.com.au
- Mon - Fri: 9:00 - 18:30
We are your trusted Australian TGA Sponsor – guiding overseas manufacturers through regulatory approval and ARTG inclusion with expertise, speed, and reliability.
The TGA are Australia’s government authority responsible for evaluating, assessing and monitoring
therapeutic goods such as medicines, medical devices and biologicals including administering the
Australian Register of Therapeutic Goods (ARTG). Devices must be listed on the register prior to sale
or advertising for sale in the Australian Market.


The TGA medical device sponsor must be an entity conducting business and located in Australia. The sponsor represents the overseas manufacturer and acts as the conduit between regulatory authorities, the public, and the manufacturer. They arrange for another party to import, export or manufacture medical devices.
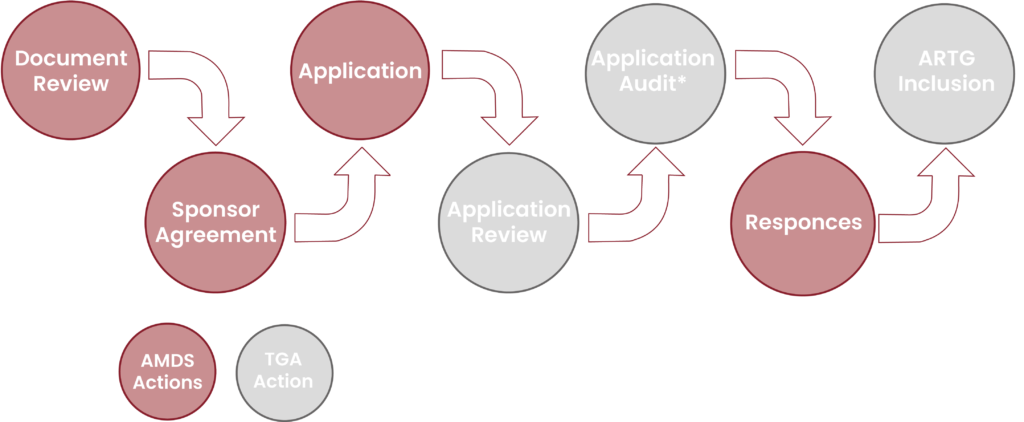
Sponsor responsibilities include.
We make the pathway into the Australian Market easy by leveraging from your existing certification and conformity assessment















Australian Medical Device Sponsors is a subsidiary of the Distinct Engineering Group.
The AMDS team of local professionals are seasoned industry specialists with combined decades of experience within the local and international medical device regulatory landscape.
Our commitment lies not only with our valued clients but also within the medical device industry, to understand your needs and provide the best possible solution and pathways into the market.
Our reach to global markets is realised through our trusted overseas partners, strategically placed within key economic jurisdictions to allow your devices access to global markets through our partner networks.
All of these attributes gives us the expertise and knowledge to manage your local interests.
Sign up for our monthly newsletter for the latest news & articles